
In the chlordecone study, liver function and structure in 32 men exposed to high levels of chlordecone while employed for 1-22 months (5.6. The subjects exposed to mirex had elevated caffeine metabolism as compared to negative control individuals (subjects with no known exposure to polyhalogenated biphenyls or other related chemicals) in which the metabolism did not increase (Lambert et al. In the mirex study, human subjects (sex and number not specified) from a chronically exposed cohort from southeast Ohio (route of exposure not specified, assumed to be oral) were assessed for cytochrome P- 4501A2 induction using a breath test that measures caffeine metabolism. Hepatic changes were observed in one chronic human exposure to mirex, as well as in a number of workers exposed to chlordecone for intermediate or chronic durations. įIGURE 4.7 Structures of the CYP1A2 substrates caffeine, phenacetin, their metabolites, and the CYP1A2 mechanism-based inhibitor, furafylline. 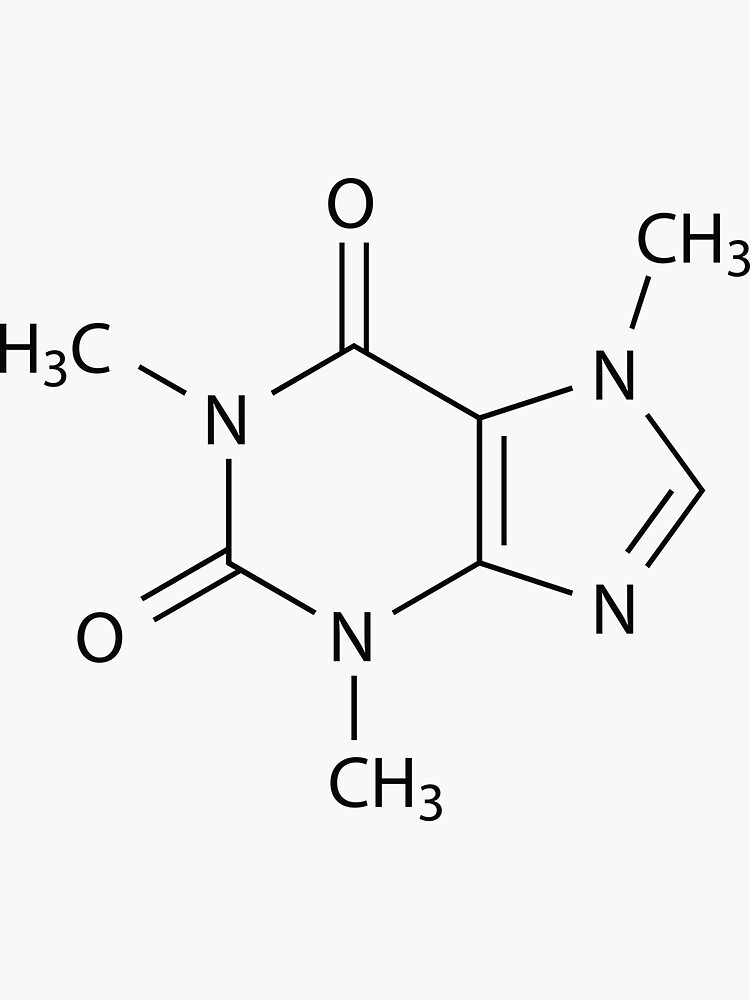 
ĭarenthin, molecular formula and structure, 5 96t Dark chocolate, 6 361 minerals content, 6 371t theobromine and caffeine content, 6 367t tocopherols, 6 370t typical formulation, 6 362t Dark conductivity ( D), of a-Si H, 22. The structures of these three are shown in Fig. This example will illustrate the quantitative analysis of aspirin ( acetylsalicylic acid), phenacetin and caffeine in a mixture. Only trace amounts of theophylline (1, 3-dimethylxanthine) are detected in cocoa and chocolate products. Caffeine (1, 3, 7-trimethylxanthine), the major purine alkaloid found in coffee and tea, is found in cocoa and chocolate at about one eighth the concentration of theobromine. Theobromine (3, 7-dimethylxanthine) is the predominant purine alkaloid in cocoa and chocolate. Structurally, they are methylated xanthines and, thus, are often referred to as methylxanthines. These include the purine alkaloids theobromine, caffeine, and theophylline. The source of cocoa beans, the species Theobroma, contains a variety of biologically active components. Each sample was a 10 mM solution in D20 loaded into the coil via the attached Teflon tubes, with the samples being (A) sucrose, (B) galactose, (C) arginine, (D) chloroquine, (E) cysteine, (F) caffeine, (G) fructose and (H).įoods derived from cocoa beans have been consumed by humans since at least 460 to 480 AD. 2.5.9 COSY spectra acquired at 600 MHz with an eight-coil probe along with the chemical structures of the compounds used. Structural formula and line drawing of caffeine, and line drawing of cholesterol. The Lewis structure of caffeine, C8H 0N4O2, a common stimulant, is shown below, (a) Give the hybridization of each atom other than hydrogen and predict the bond angles about that atom, (b) On the basis of your answers in part (a), estimate the bond angles around each carbon and nitrogen atom. Ĭomplete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicated atoms. Ĭaffeine, structure, 55, 135 Calculations, see Theoretical calculations Carbaceph-2-em, 4/3-methyl-, synthesis via l-azetin-4-one, 58, 176 Carbanions. 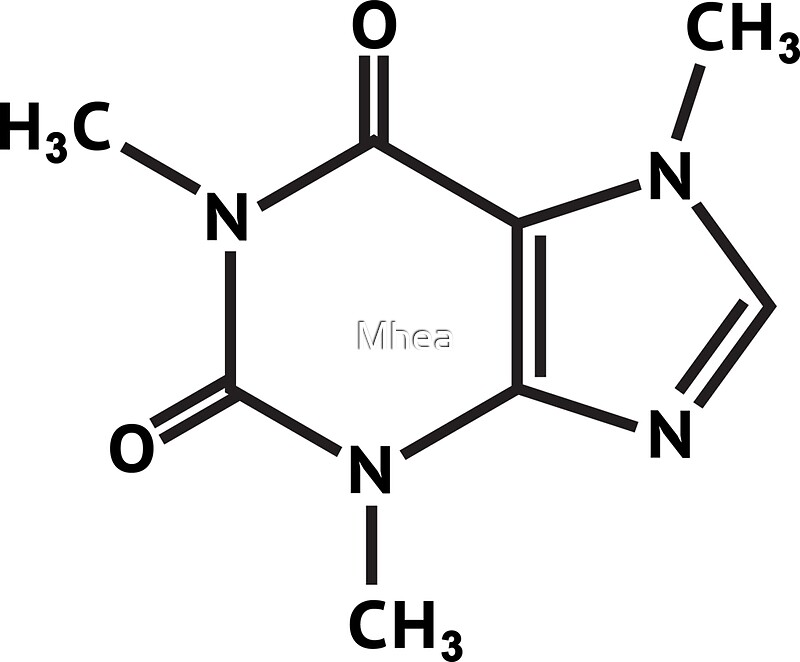
In addition, the molecule has the purine ring system, a framework which plays an important role in living systems. It is classed as an alkaloid, meaning that with the nitrogen present, the molecule has base characteristics (alkali-like).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
